A Review of Mineral Absorption with Special Consideration of Chelation as a Method to Improve Bioavailability of Mineral Supplements
By Integrative Practitioner Staff
by James Gerber, MD
Mineral absorption from supplements has been a hotly contested issue in the supplement industry. Companies naturally desire to provide the best products and gain market share by promoting them as the best. This has led many companies to claim superior absorption from particular formulations of mineral supplements. Understanding and validating some of these claims has often been difficult, due to insufficient or conflicting research. This white paper will attempt to shed some light on the controversy and establish the state of scientific knowledge about mineral absorption from supplements.
Physiology of Mineral Digestion and Absorption
In order to understand the ability of different mineral supplements to enhance mineral bioavailability, a brief review of the physiology of digestion and absorption as it impacts minerals would be useful. Since most research on mineral absorption has been done on iron, calcium and zinc, our understanding of this topic is limited primarily to these minerals.
Stomach digestion
The first significant stage of digestion occurs in the stomach, wherein hydrochloric acid (HCL) and the protein-digesting enzyme pepsin are secreted. This stage of digestion impacts mineral absorption in more ways than one.
-
Many minerals (as well as some vitamins) in the diet are bound to proteins, and protein digestion in the stomach is a necessary step in releasing the minerals from the proteins they are bound to. HCL assists protein digestion by causing protein molecules to unfold and separate from other food components, and by activating the digestive activity of pepsin.
-
HCL also directly affects minerals by helping the separation process called dissociation, wherein the positively charged mineral ion (e.g. Mg++, Ca++, Fe++, etc) becomes free from the food component or supplement compound (e.g. MgO, CaCO3, FeSO4) that it was part of.
Low HCL levels in the stomach occur frequently in the elderly, in people with some types of stomach disorders, and when any of several types of antacid drugs are used. This low HCL can prevent minerals from separating from food or supplements. If mineral separation has already occurred, low HCL can allow the minerals to recombine into compounds that are difficult to absorb (more about this in another paragraph below).
Not all mineral compounds are similarly affected by low HCL, however.
-
Iron has two possible ion states, ferrous and ferric, and ferric compounds are much less well absorbed when HCL is low compared to ferrous compounds.1 For this reason, most iron supplements are made with ferrous iron (e.g. ferrous sulfate, ferrous fumarate, amino acid chelates, etc).
So it appears that adequate HCL is often, but not always, required for optimum mineral absorption.
Intestinal digestion and absorption
After food or dissolved supplements leave the stomach, additional digestive juices are secreted into the mix by the pancreas and gallbladder. The pancreas mainly secretes digestive enzymes that further break down proteins, starches and fats, and secretes bicarbonate that neutralizes the stomach acid so that these enzymes will function optimally. Whereas HCL may have an important role in mineral absorption, it is unclear whether pancreatic enzymes play any significant part in this process.
The neutralization of stomach acid by pancreatic secretions into the intestine could have negative consequences on mineral absorption because without the influence of acid, some minerals may not remain dissociated and may bind with other components present in the intestinal contents, rendering them unavailable for absorption.7 8 9 10 11 One way this problem can be avoided is for the mineral to be surrounded by “ligands,” or weak binding agents, which will protect the mineral from stronger binding agents, even in the absence of acid, yet allow normal absorption to occur. Table 1 summarizes the effects of known binding agents found in the diet.
Table 1. Dietary ligands (weak binding agents) and their affect on mineral absorption12
|
Improve absorption (minerals affected) |
Impair mineral absorption (minerals affected) |
|
Citrate (iron, zinc, copper, manganese) |
Phytate* (calcium, magnesium, iron, zinc, selenium, chromium, manganese) |
|
Ascorbate (iron) |
Oxalate** (calcium, iron, zinc, manganese) |
|
Other organic food acids (iron, copper) |
Tannate*** (iron, zinc) |
|
Cysteine-containing peptides (iron, zinc, copper) |
Fatty acids (calcium, magnesium) |
|
Histidine and other amino acids (zinc, copper, chromium, manganese) |
|
|
Heme (iron) |
* Found in whole grains, legumes and nuts
** Found in spinach and some other vegetables, berries, nuts, tea, and chocolate
*** Found in tea and coffee
As the above table implies, dietary binding agents do not similarly affect all minerals. Notably, several binding agents may impair calcium or magnesium absorption, but agents that improve absorption of these two minerals have not been identified. To reiterate, binding agents that improve mineral absorption do so usually because they protect the mineral from making stronger attachments to other binding agents that would impair absorption, yet their weaker binding energy allows the mineral to free itself when absorption is imminent.
In addition to helpful ligands typically present in the diet, others may be secreted in digestive juices. Picolinic acid (picolinate) is a ligand found in pancreatic secretions that appears to facilitate zinc absorption, and has become a popular ingredient in some mineral formulations for this reason.13 14
The body limits mineral absorption
The body often controls mineral absorption into the intestinal wall and from there into the blood circulation. This is done in order to allow increased absorption in times of deficiency or greater need, but also to limit absorption to prevent overloading the body with minerals that can be toxic or might disturb carefully regulated blood concentrations. This control exerted by the body often places absolute limits on the percentage absorption that is possible for different minerals. For example, growing children can absorb up to 60% of their dietary calcium, whereas adults average only about 30% calcium absorption. The ability to increase supplemental mineral absorption by designing “optimal” formulations can only succeed within the limits set by these body controls. The following table shows typical mineral absorption levels at usual intakes by non-deficient people, and formulation factors that may help optimize absorption.
Table 2. Typical mineral absorption and optimizing factors12 15 16
| Mineral | Absorption | Optimizing factors |
|
Calcium |
25-35% |
Vitamin D |
|
Magnesium |
21-27% |
Vitamin D |
|
Iron (non-heme) |
5-10% |
Vitamin C, ligands |
|
Zinc |
33-41% |
Ligands |
|
Copper |
30-50% |
Ligands |
|
Selenium |
50-80% |
Incorporation into selenomethionine |
|
Chromium |
0.4-2.5% |
Vitamin C, ligands |
|
Manganese |
1-3.5% |
?? |
Supplement Formulation Effects on Mineral Absorption
In the nutritional supplement industry, many claims are made for the superior absorption of certain, sometimes proprietary, mineral formulations. To understand the issues debated concerning these claims, it is necessary to answer the question, “What is necessary for optimum mineral absorption from a supplement?”
-
At a minimum the tablet or capsule must disintegrate properly to release the mineral compound in time for proper digestion and absorption. Studies of enteric-coated (timed-release) supplements that have delayed disintegration suggest they are more poorly absorbed compared to immediate release formulations.17 18 19 20 Solubility of the mineral compound is usually assumed to be essential to absorption. However, solubility is typically measured outside of the complex gastrointestinal environment (this is called an ex vivo measurement), often by simply testing the compound in plain water or slightly acidic solutions. These simplified methods may have little relevance to what occurs during gastrointestinal processing of mineral compounds; while some studies find a correlation between solubility and absorbability of some mineral supplements, 21 others do not.22 23
-
Ionization, or dissociation, which is the separation of the mineral ion from the mineral compound (e.g. Fe+2 from FeSO4 or Ca+2 from CaCitrate), may or may not occur when a soluble mineral compound dissolves in the digestive tract. In fact, it may not be desirable for such ionization to occur. This is because a free mineral ion is vulnerable to binding with ligands, such as phytate or oxalate, that may impair absorption of the mineral, as described above (see Table 1, right column). On the other hand, if a mineral was compounded with a weak dietary ligand known to improve absorption of that mineral (see Table 1, left column), presumably by protecting it from stronger binding agents, then it would be advantageous for this compound to remain intact until the absorptive region of the intestine was reached. Unfortunately, whether mineral compounds in supplements remain non-ionized through the stomach phase of digestion has not been adequately investigated. One existing ex vivo study determined that magnesium citrate, tested in acidic conditions resembling stomach digestion, remained 65% intact as a non-ionized complex after dissolving.21 So it may be that certain mineral compounds, such as citrates, have improved absorption in part due to their ability to remain in the form of soluble complexes until absorption occurs, but more research is needed to confirm this.
The question of chelates
“Chelated” minerals are among the mineral supplements touted for their improved absorption. Before exploring the validity of such claims, the inconsistent use of the term chelate must be addressed.
Science defines chelation more precisely than does the supplement industry. The scientific definition of chelate is “organic chemicals that form two or more coordination bonds with a central metal ion. Heterocyclic rings are formed with the central metal atom as part of the ring. Some biological systems form metal chelates, e.g., the iron-binding porphyrin group of hemoglobin and the magnesium-binding chlorophyll of plants.” 24
Below is an illustration of chelates, the most relevant of which is [Fe(edta)],2- a chelate of two molecules of edta surrounding a single atom of iron.
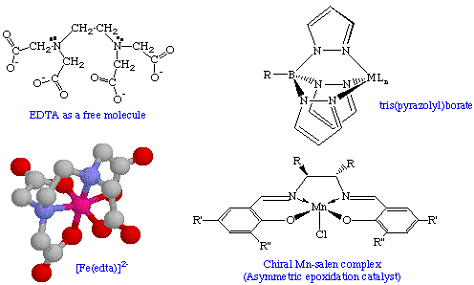
The National Nutritional Foods Association (NNFA) adopted a similar definition in 1996, specifically to establish a standard for chelated minerals using amino acids as the organic binding agents: “Metal Amino Acid Chelate is the product resulting from the reaction of a metal ion from a soluble metal salt with amino acids with a mole ratio of one mole of metal to one to three (preferably two) moles of amino acids to form coordinate covalent bonds. The average molecular weight of the hydrolyzed amino acids must be about 150 AMU (Atomic Mass Units) and the resulting chelate must not exceed 800 AMU. The minimum elemental metal content must be declared. It will be declared as a METAL amino acid chelate: e.g. Copper amino acid chelate 25
Below is an example of a reaction that results in a chelate of one atom of copper with two molecules of glycine:
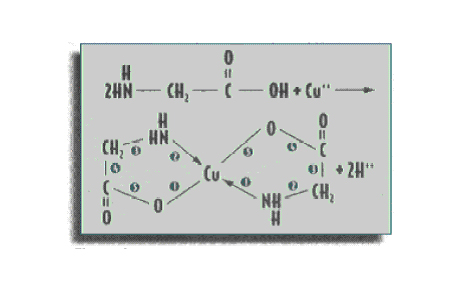
In contrast to the definitions above, dietary supplement industry often uses the term chelate to refer merely to any mineral compound formulated with amino acids or other organic acids, whether or not the compounds have been proven with special analytical tests to possess the scientific properties of a chelate.
Assuming that a mineral supplement is a true chelate, what is the evidence that this form is superior to other mineral supplements? The foremost proponent of the superiority of true mineral amino acid chelates, is Albion Laboratories of Clearfield, Utah, which develops, patents, and markets these chelates as ingredients for dietary supplements and fortified foods.26 Albion claims that the superiority of mineral amino acid chelates lies primarily in the improved absorption of such molecular configurations over other mineral compounds. This improved absorption is thought to occur by one or more theoretical mechanisms:27
-
Mineral amino acid chelates might not dissociate into free mineral ions in the stomach or intestine, preventing interactions with inhibitory dietary ligands and binding agents such as phytate and oxalate. Also, an undissociated compound may be less likely to cause gastrointestinal irritation in sensitive people.
-
Mineral amino acid chelates might be absorbed intact by amino acid absorption pathways, which allow higher percentages of absorption than the usual mineral absorption pathways.
Either or both of the mechanisms described above could help minimize the competition for absorption known to occur between certain pairs of minerals when they are both present in the intestine.
Research on iron amino acid chelate
The absorption and nutritional value of mineral amino acid chelates has not been scientifically studied in humans, with one exception – Ferrochel – a patented Albion product with the chemical name iron bis-glycine chelate. The handful of published human studies on Ferrochel gives some indication of the potential for chelated minerals to deliver benefits not possible with other mineral compounds.
Most tests of Ferrochel absorption have compared it to ferrous sulfate, the most common dietary iron supplement. Moreover, these absorption studies were typically carried out by mixing the iron compounds with various foods, in order to test the value of the compounds as ingredients of fortified grain products, infant formulas, etc. According to unpublished research reports by Albion, Ferrochel was absorbed up to five times better than ferrous sulfate when given to adult males in a cornmeal porridge, and 59% better when given to adults as tablets.27 Published research has reported Ferrochel to be about two times better absorbed than ferrous sulfate when given in bread to adults, some of whom were iron-deficient,28 and about four times better absorbed when given to adult males in a cornmeal porridge.29 However, no difference between these iron sources were found when used in weaning foods for 9-month old infants,30 and no difference in absorption between Ferrochel and ferrous ascorbate was found when the compounds were given in water to adult women.31
Albion claims that chelated iron is absorbed by a pathway different from how other forms of iron are absorbed. However, a recent study showed that iron bis-glycine chelate (not identified as Ferrochel) influenced the absorption of ferrous sulfate in a way suggesting that iron chelates are absorbed by typical non-heme iron pathways.32 The investigators concluded that iron chelates are more bioavailable than other iron forms because they are protected from absorption inhibitors that are typically present in the intestine when iron compounds are mixed with food.
From the above evidence, it appears that the primary benefit of mineral amino acid chelates may be to improve absorption when these compounds are used in or taken with foods containing absorption inhibitors. Used in water, no advantage of chelates was found, but unfortunately no absorption research involving tablets or capsules taken away from meals has been published. However, another avenue of human research has examined the treatment of iron-deficiency anemia with chelated iron versus ferrous sulfate supplements. Anemic and malnourished infants and young children given Ferrochel in syrup for four weeks had similar improvements in hemoglobin levels but better improvements in levels of stored iron compared to infants receiving ferrous sulfate.33 Apparently, Ferrochel allowed more rapid restoration of iron stores during the short four-week treatment. In another trial, anemic adolescents were given tablets to supply 30, 60, or 120 mg/day of iron from Ferrochel or 120 mg of iron from ferrous sulfate.34 These tablets were to be taken either with breakfast or supper. At the end of four weeks, hemoglobin was similarly improved in all groups while ferritin improved in all but the group receiving the lowest amount of Ferrochel. These investigators concluded that lower doses of Ferrochel obtained similar results to higher doses of ferrous sulfate over the short-term treatment. While these studies suggest anemic people may respond faster to chelated iron than to ferrous sulfate, there remains no evidence that chelated iron is superior to forms of iron other than the sulfate, or that chelated iron would perform better when taken in tablets or capsules away from food.
References
1. Champagne ET. Low gastric hydrochloric acid secretion and mineral bioavailability. Adv Exp Med Biol 1989;249:173-84.
2. Aymard JP, Aymard B, Netter P, et al. Haematological adverse effects of histamine H2-receptor antagonists. Med Toxicol Adverse Drug Exp 1988;3:430-48.
3. Sturniolo GC, Montino MC, Rossetto L, et al. Inhibition of gastric acid secretion reduces zinc absorption in man. J Am Coll Nutr 1991;10:372-5.
4. Recker RR. Calcium absorption and achlorhydria. N Engl J Med 1985;313:70–3.
5. Bo-Linn GW, Davis GR, Buddrus DH, et al. An evaluation of the importance of gastric acid secretion in the absorption of dietary calcium. J Clin Invest 1984;73:640–7.
6. Serfaty-Lacrosniere C, Woods RJ, Voytko D, et al. Hypochlorhydria from short-term omeprazole treatment does not inhibit intestinal absorption of calcium, phosphorus, agnesium or zinc from food in humans. J Am Coll Nutr 1995;14:364–8.
7. Heijnen AM, Brink EJ, Lemmens AG, Beynen AC. Ileal pH and apparent absorption of magnesium in rats fed on diets containing either lactose or lactulose. Br J Nutr 1993;70:747-56.
8. Heaney RP, Recker RR, Weaver CM. Absorbability of calcium sources: the limited role of solubility. Calcif Tissue Int 1990;46:300-4.
9. Rumenapf G, Schwille PO. The influence of citrate on the duodenal absorption of calcium in the rat. Calcif Tissue Int 1988;42:326-30.
10. Hungerford DM Jr, Linder MC. Interactions of pH and ascorbate in intestinal iron absorption. J Nutr 1983;113:2615-22.
11. Seal CJ, Heaton FW. Chemical factors affecting the intestinal absorption of zinc in vitro and in vivo. Br J Nutr 1983;50:317-24.
12. Groff JL, Gropper SS. Advanced nutrition and human metabolism, 3rd ed. Belmont, CA: Wadsworth, pp. 371-483.
13. Ijuin H. Evaluation of pancreatic exocrine function and zinc absorption in alcoholism. Kurume Med J 1998;45:1-5.
14. Barrie SA, Wright JV, Pizzorno JE, et al. Comparative absorption of zinc picolinate, zinc citrate and zinc gluconate in humans. Agents Actions 1987;21:223-8.
15. Shils ME, Olsen JA, Shike M, Ross AC. Modern nutrition in health and disease, 9th edition. Baltimore: Williams & Wilkins, 1999.
16. Food and Nutrition Board, Institute of Medicine. Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. Washington DC: National Academy Press, 2002.
17. Fine KD, Santa Ana CA, Porter JL, Fordtran JS. Intestinal absorption of magnesium from food and supplements. J Clin Invest 1991;88:396-402.
18. Ricketts CD. Iron bioavailability from controlled-release and conventional iron supplements. J Appl Nutr 1993;45:13–19.
19. Rudinskas L, Paton TW, Walker SE. Poor clinical response to enteric-coated iron preparations. Can Med Assoc J 1989;141:565–6.
20. Walker SE, Paton TW, Cowan DH, et al. Bioavailability of iron in oral ferrous sulfate preparations in healthy volunteers. Can Med Assoc J 1989;141:543–7.
21. Lindberg JS, Zobitz MM, Poindexter JR, Pak CY. Magnesium bioavailability from magnesium citrate and magnesium oxide. J Am Coll Nutr 1990;9:48-55.
22. Heaney RP, Recker RR, Weaver CM. Absorbability of calcium sources: the limited role of solubility. Calcif Tissue Int 1990;46:300-4.
23. Hansen C, Werner E, Erbes HJ, et al. Intestinal calcium absorption from different calcium preparations: influence of anion and solubility. Osteoporos Int 1996;6:386-93.
24. Hawley’s Condensed Chemical Dictionary, 12th ed.
25. Adopted by the NNFA Board of Directors, July 1996. NNFA Today, August 1996, p. 15.
27. Ashmead HD. The absorption and metabolism of iron amino acid chelate. Arch Latinoam Nutr 2001;51(1 Suppl 1):13-21.
28. Layrisse M, Garcia-Casal MN, Solano L, et al. Iron bioavailability in humans from breakfasts enriched with iron bis-glycine chelate, phytates and polyphenols. J Nutr 2000;130:2195-9.
29. Bovell-Benjamin AC, Viteri FE, Allen LH. Iron absorption from ferrous bisglycinate and ferric trisglycinate in whole maize is regulated by iron status. Am J Clin Nutr 2000;71:1563-9.
30. Fox TE, Eagles J, Fairweather-Tait SJ. Bioavailability of iron glycine as a fortificant in infant foods. Am J Clin Nutr 1998;67:664-8.
31. Olivares M, Pizarro F. Bioavailability of iron bis-glycinate chelate in water. Arch Latinoam Nutr 2001;51(1 Suppl 1):22-5.
32. Pizarro F, Olivares M, Hertrampf E, et al. Iron bis-glycine chelate competes for the nonheme-iron absorption pathway. Am J Clin Nutr 2002;76:577-81.
33. Pineda O, Ashmead HD. Effectiveness of treatment of iron-deficiency anemia in infants and young children with ferrous bis-glycinate chelate. Nutrition 2001;17:381-4.
34. Pineda O, Ashmead HD, Perez JM, Lemus CP. Effectiveness of iron amino acid chelate on the treatment of iron deficiency anemia in adolescents. J Appl Nutr 1994; 46:2-11.















