FDA pain draft promotes alternative medicine, offers chance for influence
By John Weeks
 by John Weeks, Publisher/Editor of The Integrator Blog News and Reports
by John Weeks, Publisher/Editor of The Integrator Blog News and Reports
The Food and Drug Administration (FDA) released proposed changes Wednesday to its blueprint on educating health care providers about treating pain. The guidelines now recommend that doctors get information about chiropractic care and acupuncture as therapies that might help patients avoid prescription opioids.
When I originally received the e-mail with the news, the only comment from the sender of the email was a quote from a young Bob Dylan: “The times they are a changin’.”
Indeed, they are—sort of. Readers will have a chance to help hurry those changes in pain treatment along by helping shape the FDA draft.
The agency, long lambasted by the integrative health community for its revolving door of leadership with big pharmaceuticals, released its initial version of “FDA Education Blueprint for Health Care Providers Involved in the Management or Support of Patients with Pain.” Under Section 2, part two, on how to create a treatment plan, the very first consideration urged by the FDA offers is, “Nonpharmacologic therapies– includes psychological, physical rehabilitative, surgical approaches; and complementary therapies.”
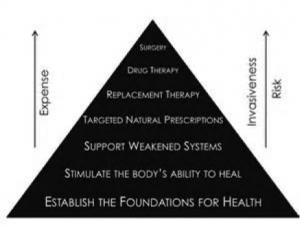 Pharma follows next, as it should in a therapeutic order that seeks to do the least harm. (See figure.)
Pharma follows next, as it should in a therapeutic order that seeks to do the least harm. (See figure.)
A subsequent section of the document on non-pharmacological approaches also lists “complementary therapies.” They are again at the tail end of the same set of considerations, following psychiatric, rehabilitation, and surgery. This is a topsy-turvy therapeutic order, with apparently little philosophy of medicine shaping it.
This section does, however, get more specific in what it means by complementary therapies. The agency gives gives acupuncture and “chiropracty” as examples. Explicit reference, even using an archaic term for chiropractic, is good for those called out.
For reference, and to raise reasonable concern, the December 2016 CDC draft guidance on opioid prescribing, for instance, originally called for “many nonpharmacologic therapies, including physical therapy, weight loss for knee osteoarthritis, complementary and alternative therapies (e.g., manipulation, massage, and acupuncture).” The March 2016 final guidance mentioned neither the category nor the specific approaches.
The FDA’s quarter page segment on non-pharma is effectively damning with faint praise as this cameo is swamped by seven pages on drug management strategies. Complementary therapies do not make another appearance in these pages, unless one is hungry enough to answer to “etc”. In a section on “Patient Education” the closest mention is, “Health care practitioners should inform patients about pain management expectations and managing pain through different modalities (non-opioids, rest, physical therapy, occupational therapy, etc.) when appropriate.”
In a section at the end on Addiction Medicine, the agency recommends that practitioners know “the types of pharmacologic and non-pharmacologic treatments available.” No specifics are offered.
The FDA has not yet set up a docket to support comments and response. See their notice here.
 Comments: There is a chance for activism here, since this was a draft. As noted, the inclusions here can be swept out of the final, as the CDC did. The FDA hasn’t yet set up the docket for responding.
Comments: There is a chance for activism here, since this was a draft. As noted, the inclusions here can be swept out of the final, as the CDC did. The FDA hasn’t yet set up the docket for responding.
The title of the STAT article clearly overplays the reality of this document. This is nowhere more evident than a recommendation buried in the many pages on opioids. Think of what you might consider if you imagine a “paradigm shift” in pain management. One would take that “non-pharma first” guidance and help practitioners and their patients in all ways possible to create the optimal use of self-care, integrative, and non-pharmacologic approaches in pain treatment to prevent the need for, and get off of, opioids.
Here, in counter position, is the FDA, referencing this concept:
“Paradigm shift in opioid prescribing – HCPs should be knowledgeable about existing information about safe opioid practices current federal and state regulations, and guidelines on opioid prescribing and the use of naloxone.”
 Nothing of complementary and alternative products, practices and practitioners in this new paradigm.
Nothing of complementary and alternative products, practices and practitioners in this new paradigm.
The community has its work cut out for it. Here are some directions for comments, when the FDA opens its docket for comments:
- If they want to see non-pharma, they need to expand the non-pharmacologic and complementary health section from a half page to at least three or more and fill it with good content. There is plenty of good content to be used if its new paradigm is to significantly include non-pharma. The document’s time and space devoted to the topic, via content such as that recommended in the bullets below, is part of the message to practitioners.
- Build out an area on patient education. Ask for help from experts in the integrative health and medicine community in doing so.
- Explicitly note other practices such as are mentioned in the recent ACP guideline on back pain. Among those they should explicitly mention are massage, self-care, mind-body, tai chi, yoga, use of integrative, naturopathic and functional medicine specialists.
- Drive health care practitioners to the ACP guidelines and the Joint Commission’s pain standard.
- Provide a section on strategies for gaining coverage of non-pharmacologic approaches. This is a very challenging area. Teach them to advocate with insurers for coverage. This is essential.
- Include more integrative services in the seven pages on managing drugs to remind prescribers that they needn’t stay lost in the pharmaceutical woods, that non-pharma, integrative approaches can both limit need for the drugs but also assist in the exit strategy.
- Talk about the importance of partnership with complementary practitioners that the National Pain Strategy has recommended.
- Teach them that most patients and practitioners don’t think of “chiropracty” when thinking of what chiropractic doctors do, so why not try using the language of the realm. What is that about?
One last reminder. Recall from the CDC guidance on opioid prescribing that inclusion in a draft does not insure inclusion in the final. The language that excited the write at STAT could be blown away by a little political pea shooting, as CDC’s draft language was rumored to have been shot down. A strong respond from integrative health practitioners and organizations is in order. Frustrating that they would issue this without having the comment docket up.















